
SCFE: Super Critical Fluid Extraction
A supercritical fluid is a state of matter where a substance is at a temperature and pressure above its critical point. At this point, the liquid and gas phases of the substance merge into one phase with unique properties. Supercritical fluids can diffuse through solids like a gas and dissolve materials like a liquid. Carbon dioxide is a common example, widely used in various applications due to its moderate critical temperature and pressure.
Supercritical fluids, particularly supercritical CO2, are used for extraction due to their unique properties. For example, supercritical fluids exhibit tunable solvency, low viscosity, and high diffusivity, and leave no residue; they are non-toxic and environmentally friendly. Amar has offered supercritical fluid extraction (SCFE) equipment for many years. We are able to design and fabricate systems ranging from pilot scale to large production units. These are extremely specialized designs with high pressure components and valves. We even have the ability to conduct a demo for your application at our on-site SCFE unit which is available for initial testing.
Supercritical CO2 is non-toxic and non-flammable, making it safer and more environmentally friendly than organic solvents. We find wide applicability from users such as Fragrances, Flavors, Medicinal extracts, spices, essential oils, and many others. Recently SCFE is also being used for textile dying applications where it converts a polluting legacy process to a clean alternative. SCFE also leaves no solvent residue in the extracted products, which is crucial for food, pharmaceuticals, and other applications with increasingly strict rules on residual solvent content. The process can be quicker and more efficient compared to traditional solvent extraction due to the enhanced solubility and diffusion properties. The Table below shows typical binary diffusion coefficients in various systems (reference mentioned later in this blog) and shows the key benefit of SCFE: Diffusion is about 10x faster than in the liquid phase.

Successful production processes include supercritical extraction, fractionation, dyeing, cleaning, degreasing, and micronization (rapid expansion, crystallization, anti-solvent recrystallization). New material properties can be achieved by foam expansion, aerogel drying, polymer processing, impregnation, and cell-cracking with high pressure supercritical CO2. (Reference: The book by Bertucco & Vetter on high pressure processes)
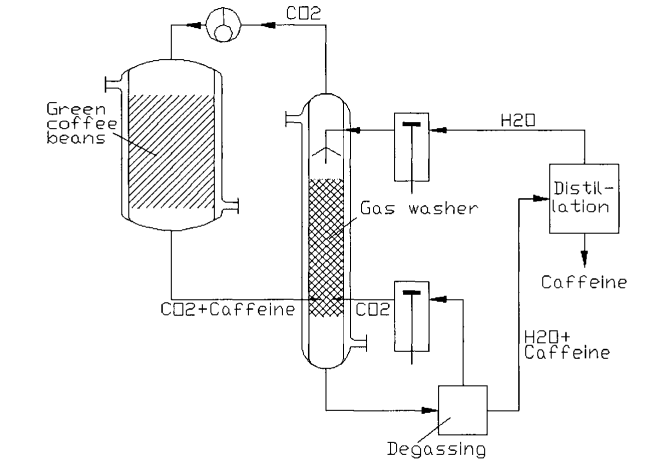
Other traditional applications of supercritical fluids include the decaffeination of coffee and tea, extraction of flavors and fragrances, pharmaceutical production, polymer processing, and environmental remediation. The figure below (Produced from the same book mentioned above) shows a typical conceptual SCFE used for the decaffeination of coffee beans. The heart of the process are the high pressure extraction columns (#5) and the phase separator (#8). The process is semi-continuous with fresh coffee beans periodically loaded in column #6. Readers are referred to the original reference for additional details.
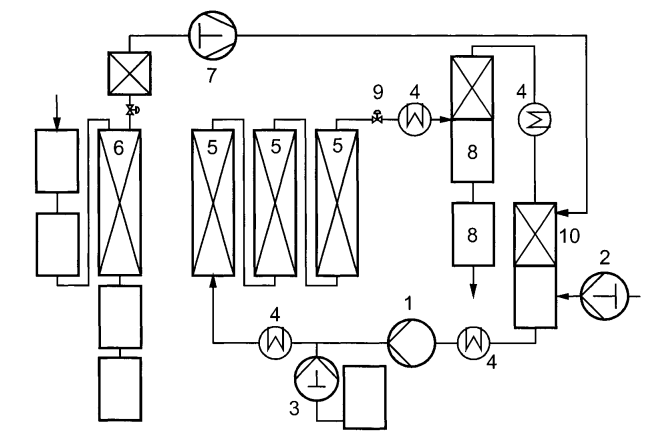
In fact, the decaffeination of caffeine was one of the pioneer processes for SCFE and the historical sketch below (reproduced from the reference above) shows one of the very first units conceptualized by Zosel.
Due to the high pressures and the complex process design, it is essential to use a vendor you trust. Key aspects in equipment selection for supercritical fluid applications include material compatibility, pressure and temperature control, safety features, ease of maintenance and cleaning, and cost-effectiveness.
Talk to us at Amar for your next SCFE requirements. [email protected]